Current Research
Spatiotemporal structure of local sleep
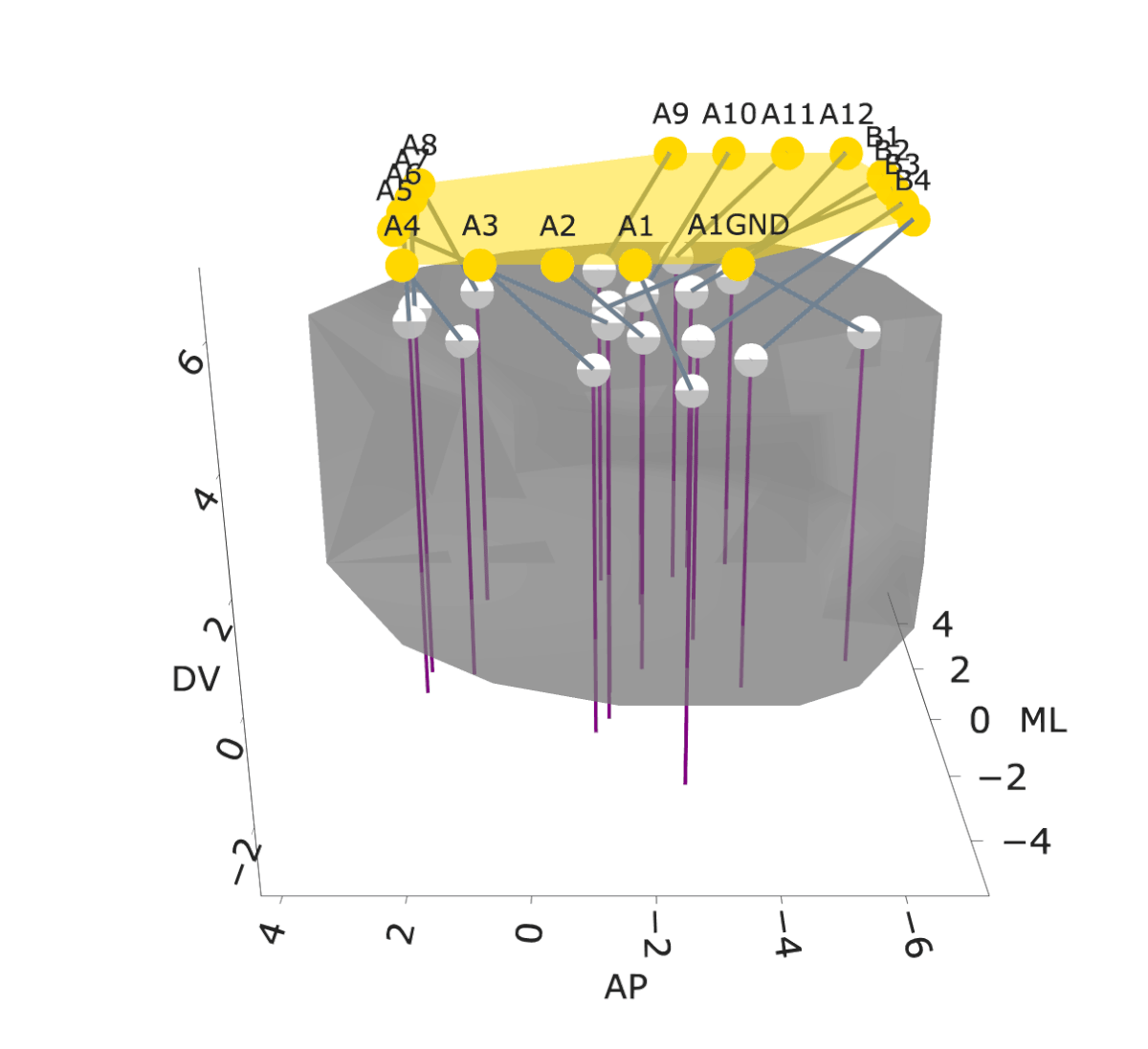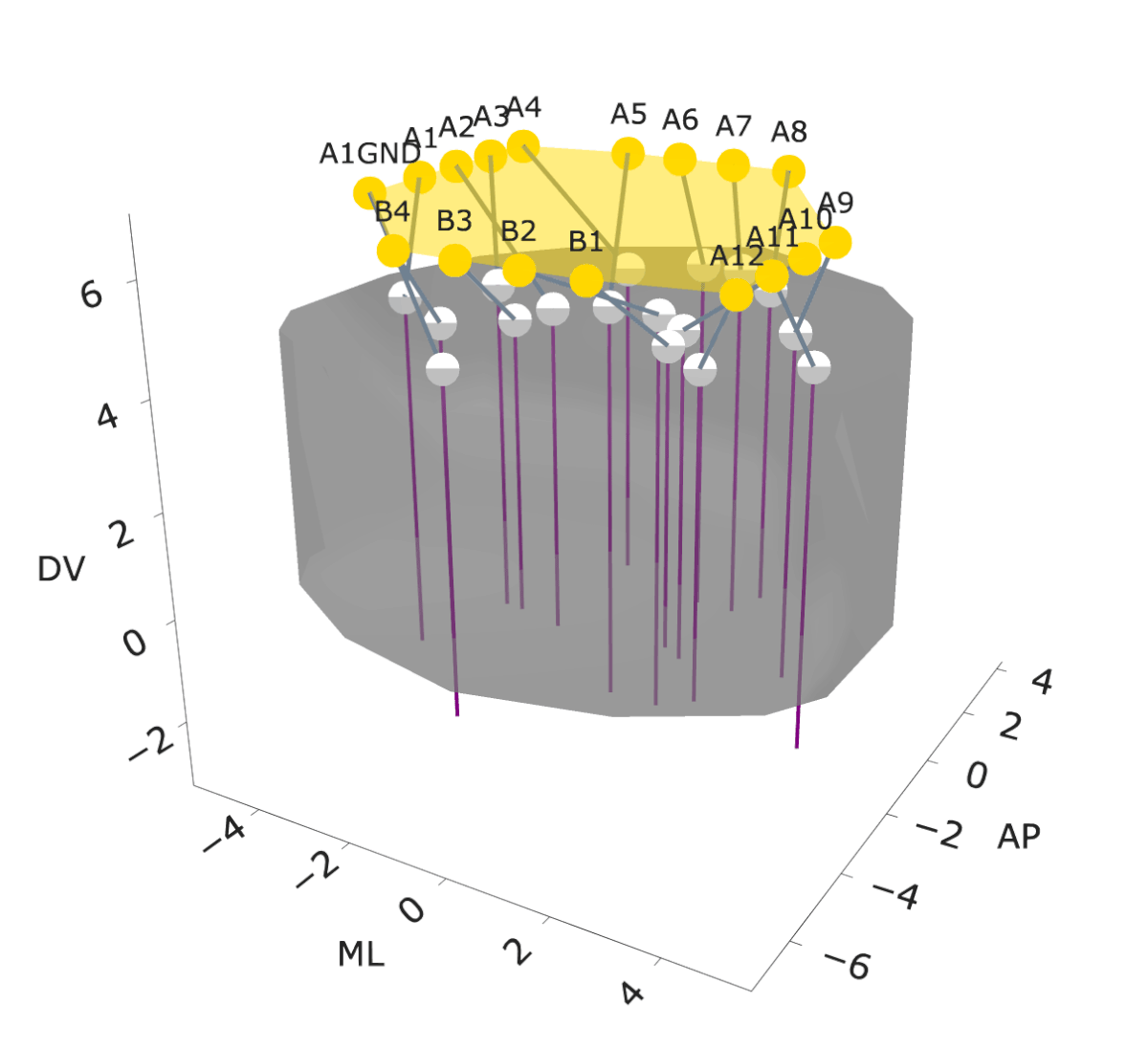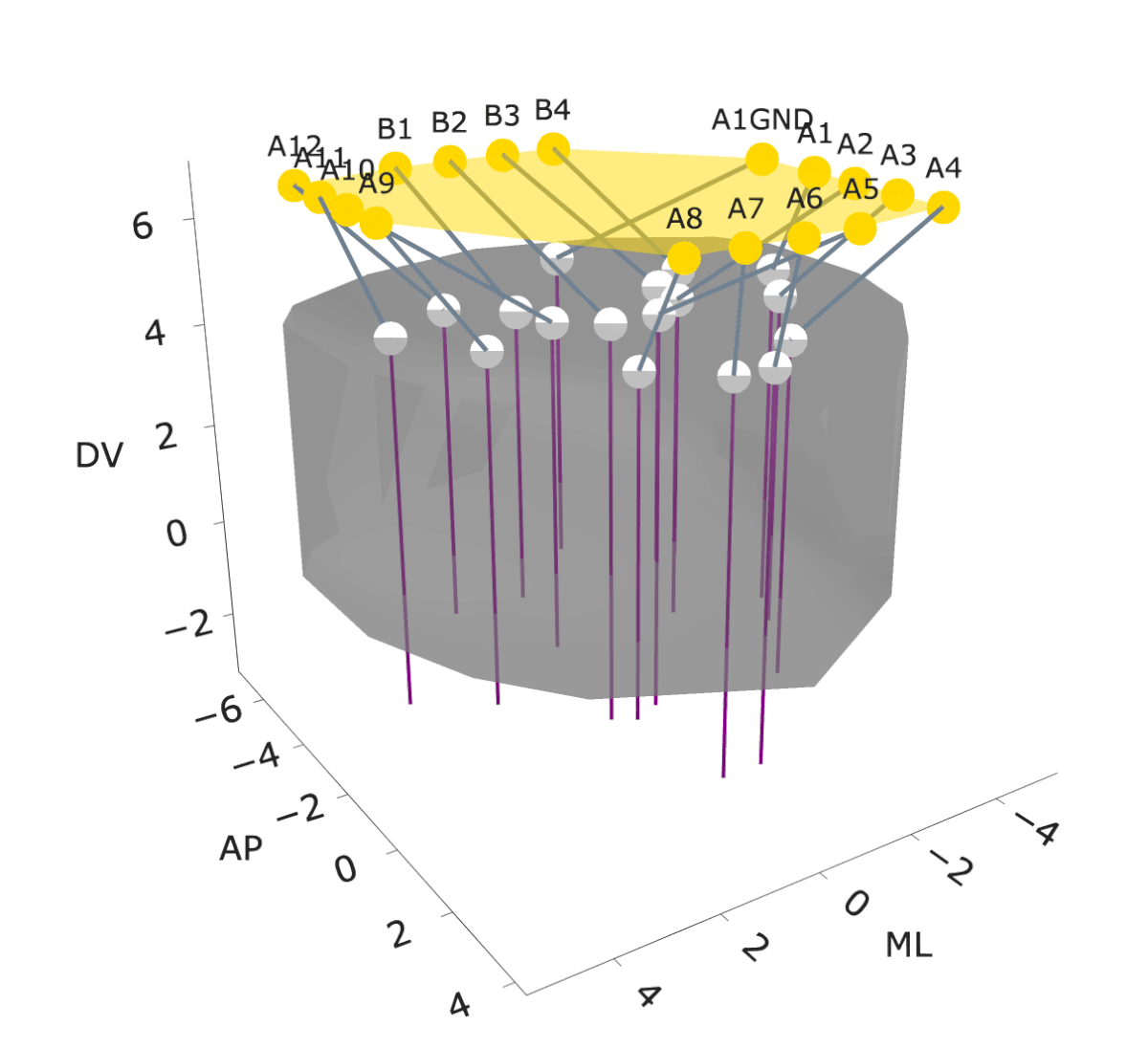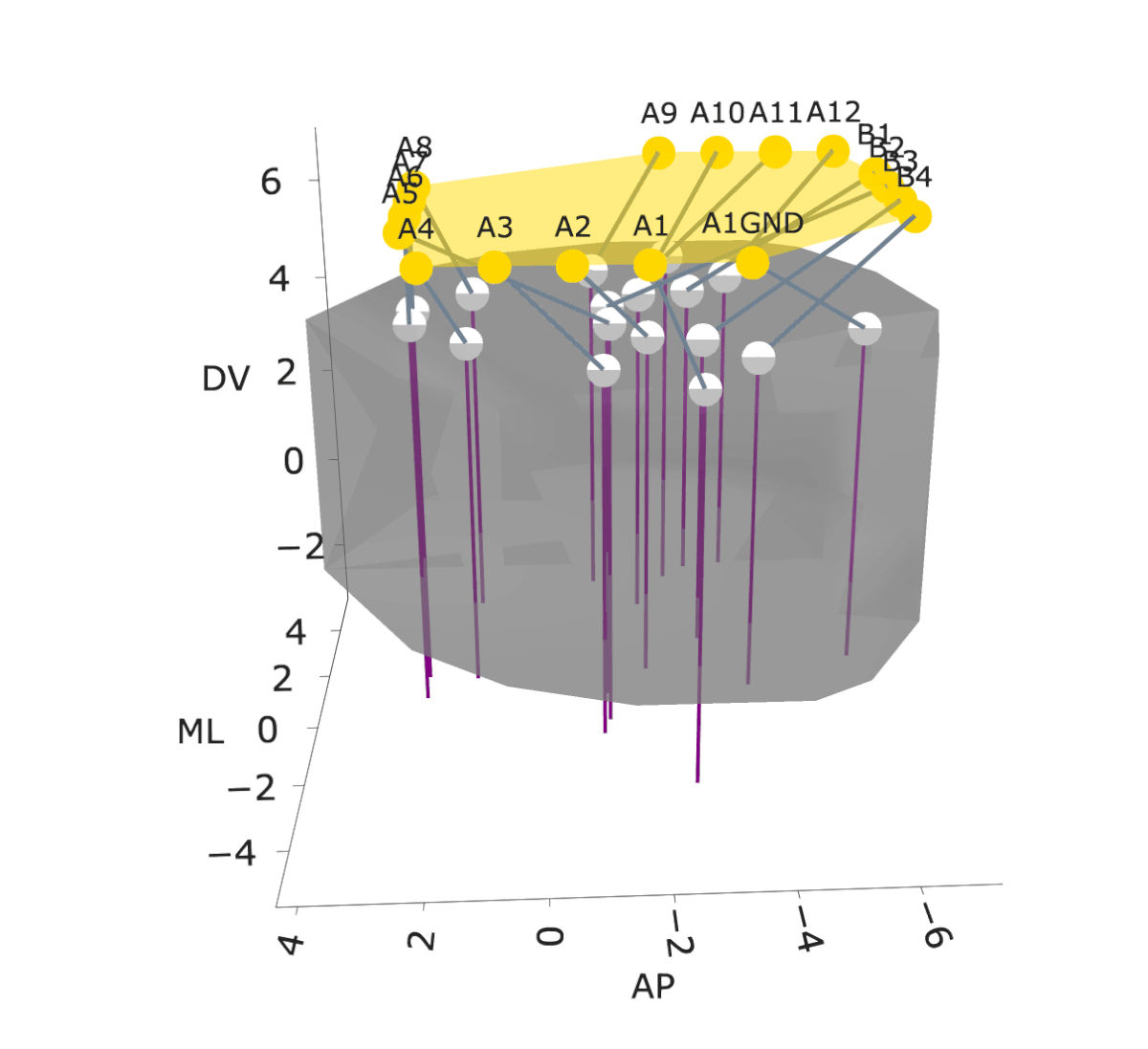
NightCap to record across 16 brain regions simultaneously
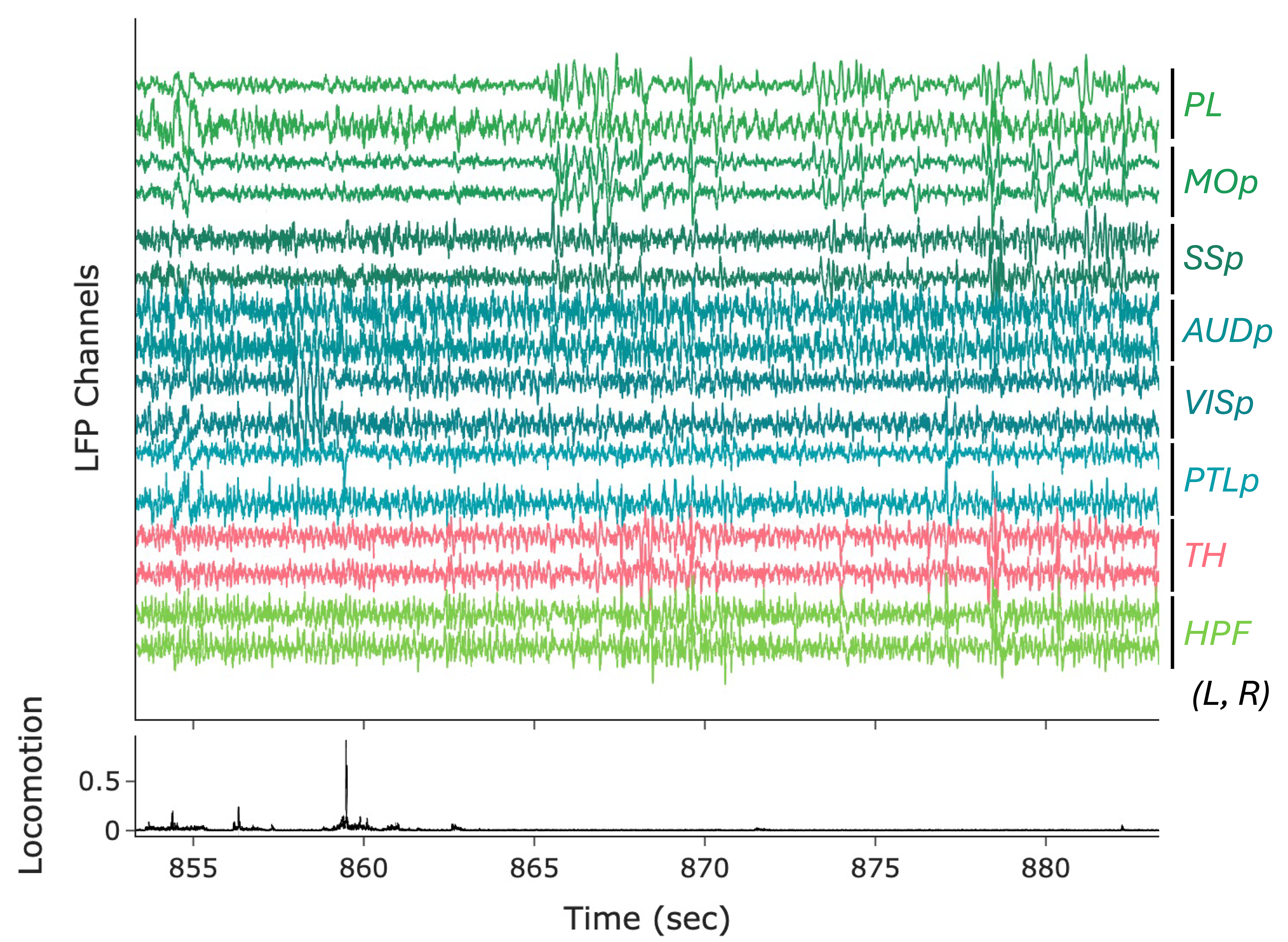
example snippet of electrophysiology across the brain

wavelet coherence across 16 brain regions
Sleep has historically been viewed as a globally uniform state—when an animal is in a state of sleep, the entire brain is in the same state. However, recent studies have discovered evidence for local sleep during which specific brain areas display electrophysiological signatures of sleep independently of the rest of the brain. Evolution has produced remarkable diversity of sleep structure across the animal kingdom, including species that sleep with only one hemisphere of their brain at a time (e.g., dolphins), species that switch between this “unihemispheric” sleep and bihemispheric sleep in an environmental-dependent manner (e.g., seals, penguins, certain birds), and species that have subtler forms of local sleep where specific regions of the brain deviate from an otherwise globally uniform state (e.g., rodents). These observations of local sleep disrupt the notion that sleep is invariably a brain-wide state.
In my postdoctoral work, I am investigating the spatiotemporal structure of local sleep across the mouse brain, and testing whether and how experience and neuronal activity during wake periods alter the structure of subsequent sleep. I am employing state-of-the-art electrophysiological recording tools, engineering solutions to record electrophysiology broadly across the mouse brain, and combining these electrophysiology tools with calcium imaging, chemogenetics, optogenetics, and behavioral assays in freely moving mice to observe and perturb neuronal dynamics to investigate how sleep is structured in space and time across the mouse brain.
Past Projects

calcium imaging schematic
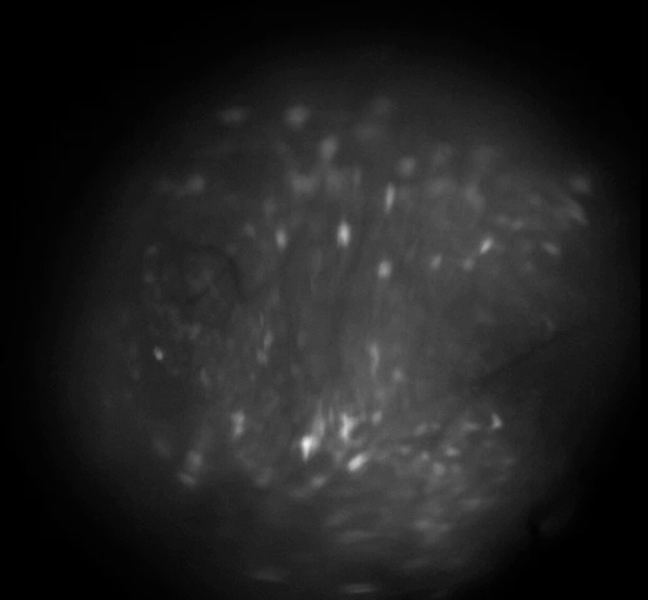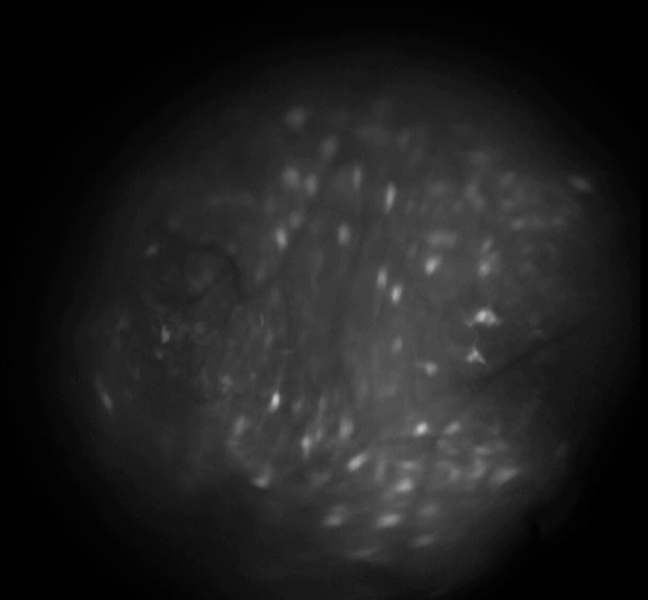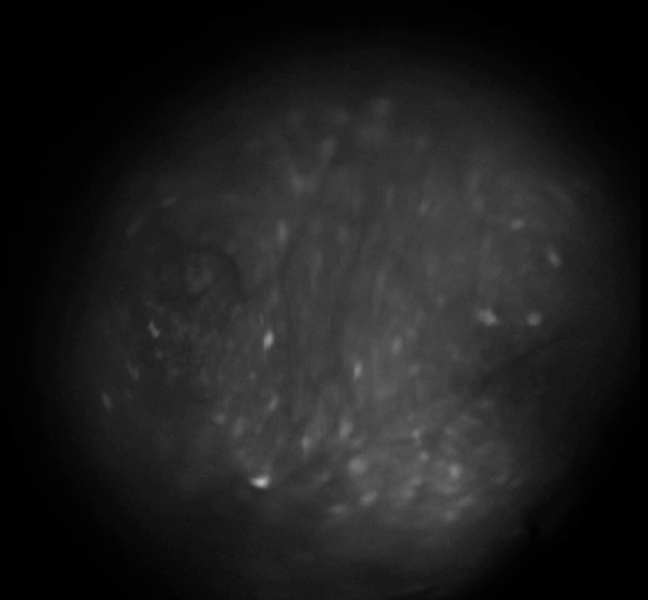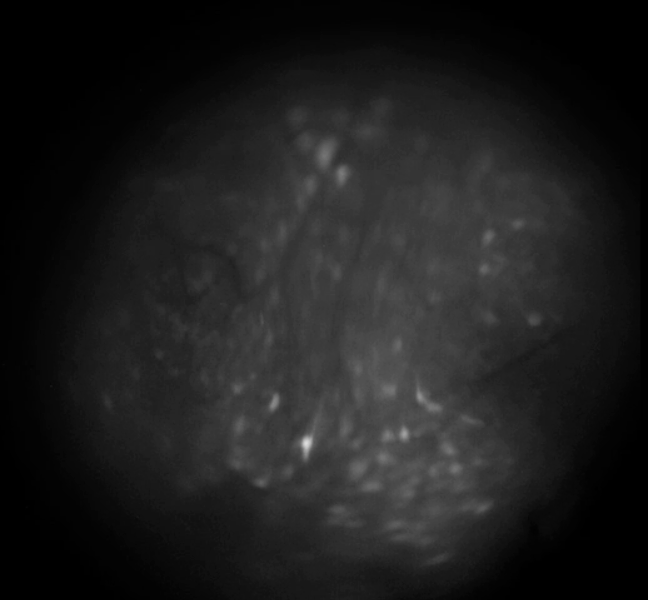
Example calcium imaging recording
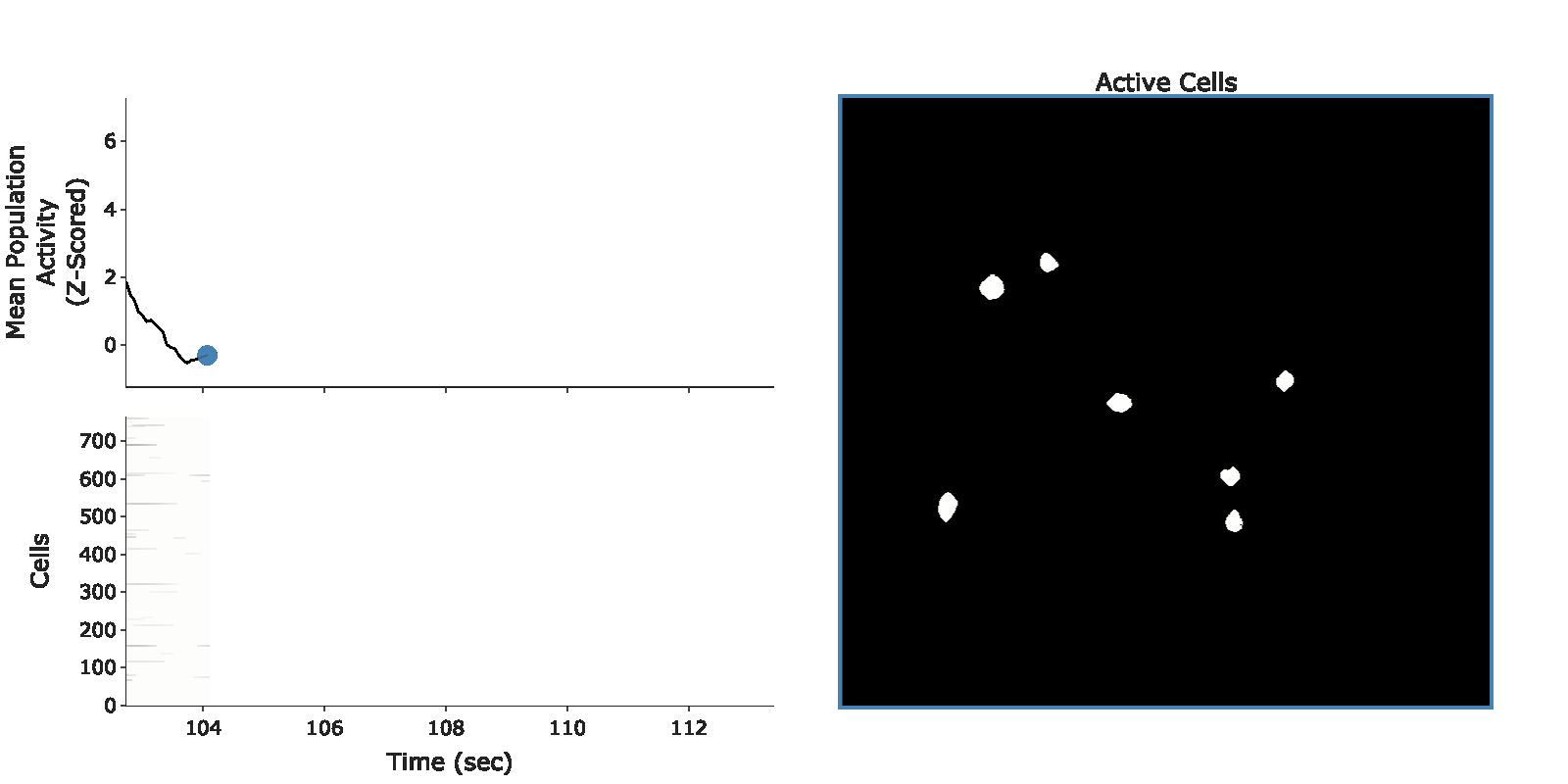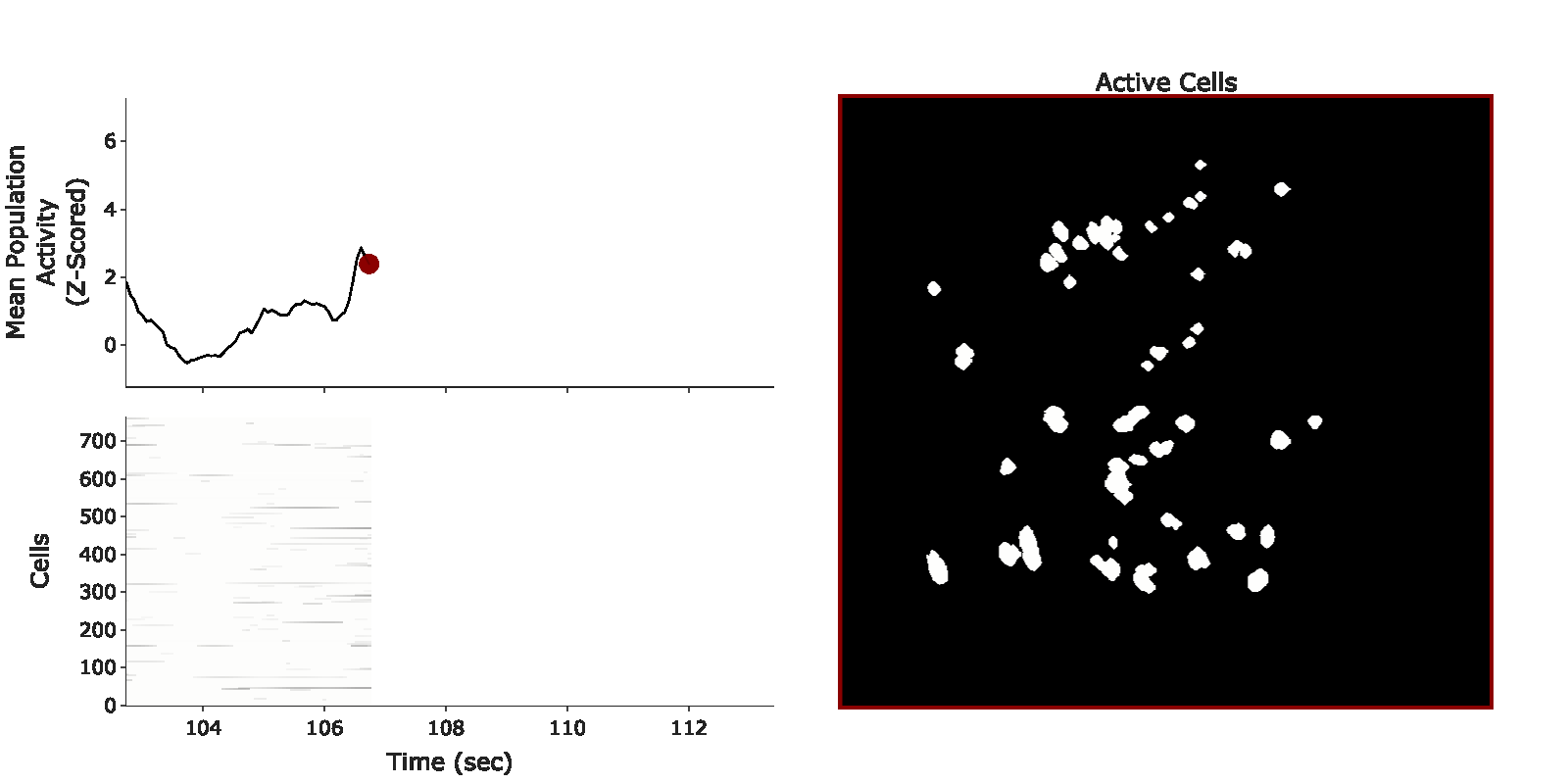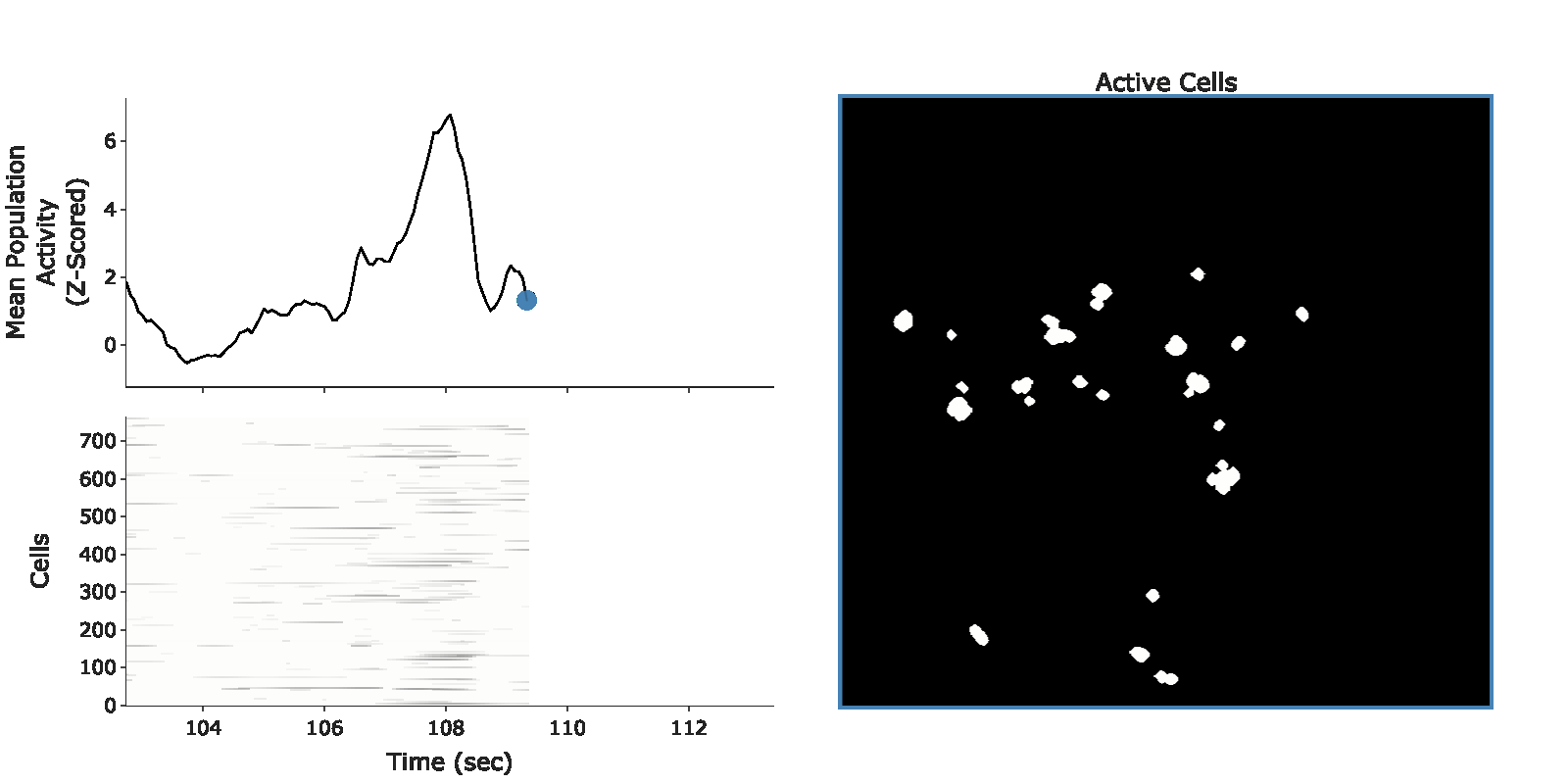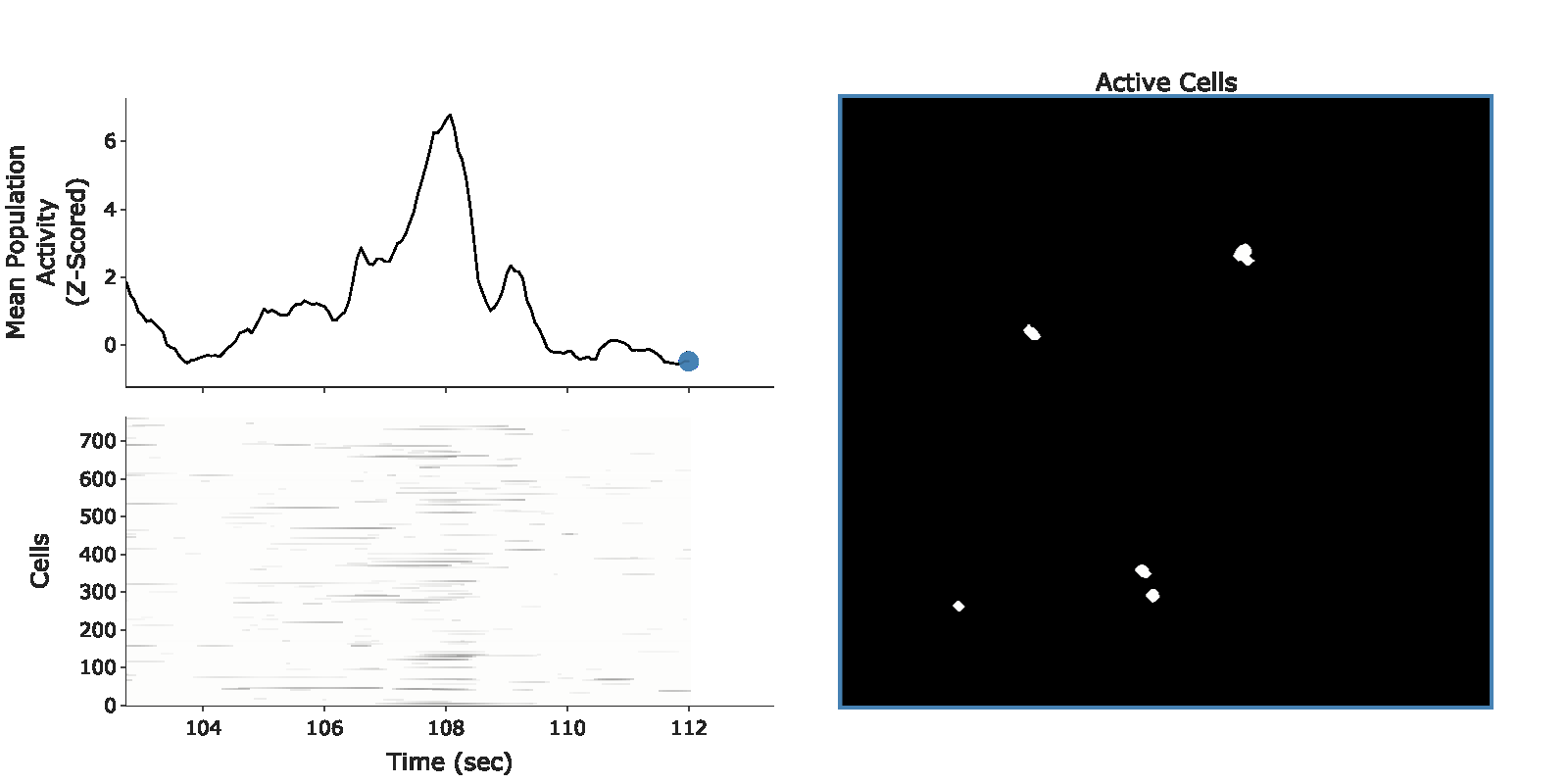
example population burst event
In my PhD, I studied how animals integrate and segregate memories across days. I found that fear from a highly aversive memory can spread to memories formed days ago—termed retrospective memory-linking. I found that retrospective memory-linking is driven by the period immediately following learning, where neuronal ensembles representing the two memories are co-reactivated, driving the formation of an integrated ensemble and, in turn, integrated memories. This work was published in Nature, and has been featured in the NIH Director's Blog, in The Transmitter, in US News, in StudyFinds, and in the Mount Sinai Newsroom.
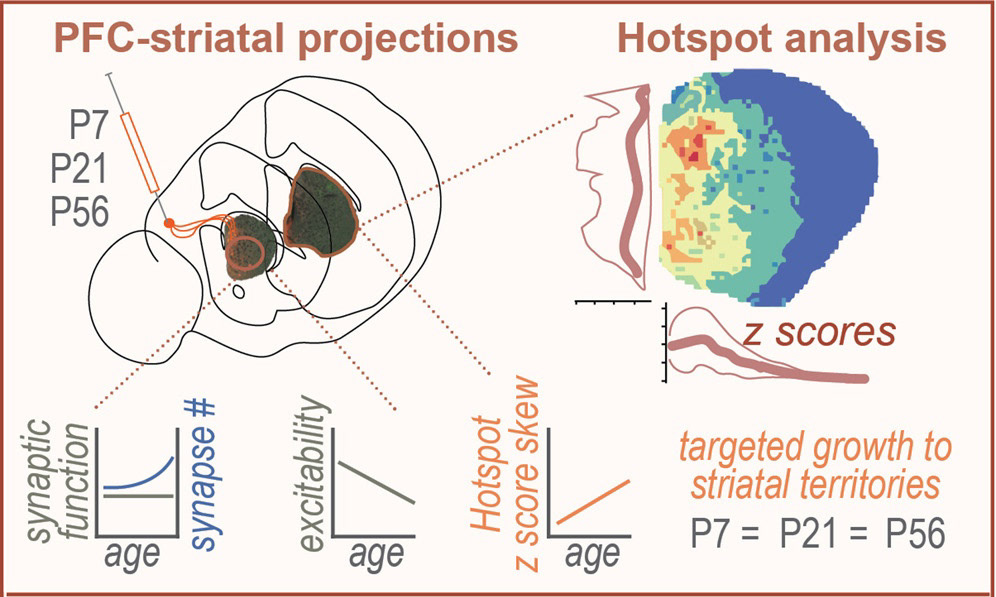
partial graphical abstract of paper
During brain development, cortical neurons send large axonal projections to several different parts of the brain. Among these projections, the striatum is a large target of many deep-layer cortical neurons. These corticostriatal projections form a topography in the striatum in such a way that different cortical areas terminate in spatially restricted regions of the striatum. How cortical growth cones know where to terminate their axons in striatum to form this topography remains poorly understood. This project sought to investigate how corticostriatal projections are formed across development, and to test the role of cadherin-8, a key cell adhesion molecule involved in synapse formation, in this process. For this project, I developed an algorithm to track the formation of innervation specificity across postnatal development, and to quantify the disruption of innervation patterns after genetic deletion of cadherin-8. This work was published in iScience.
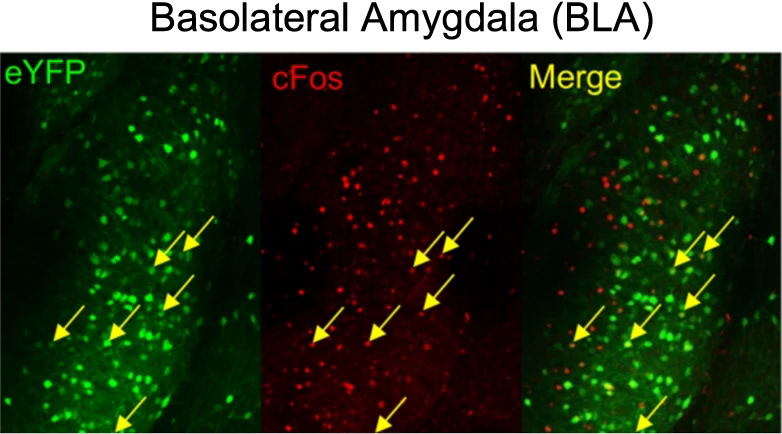
example co-localization analysis
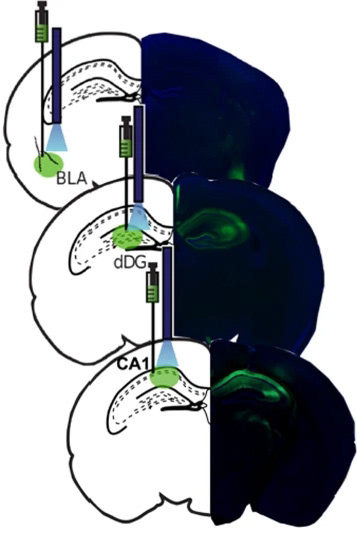
target sites for optogenetic manipulation
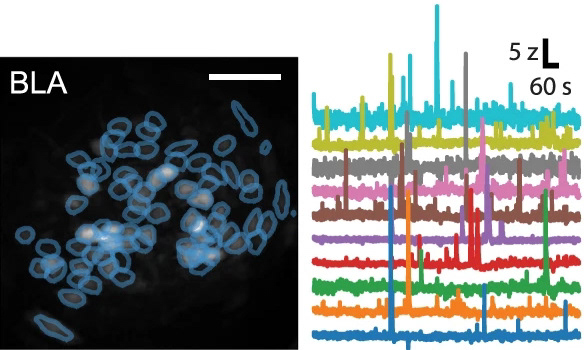
example amygdalar calcium imaging
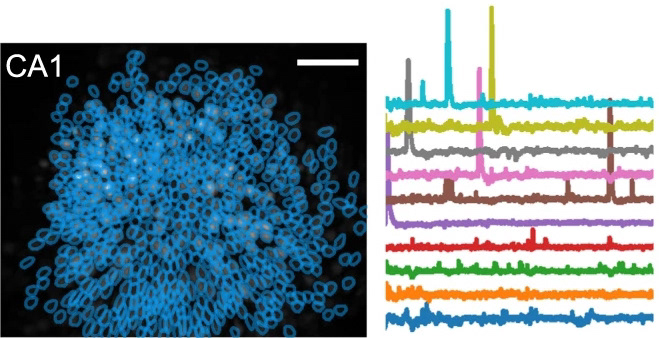
example hippocampal calcium imaging
When a fearful experience occurs in a neutral environment, the environment becomes predictive of a fearful outcome, referred to as contextual fear conditioning. This forms a fear memory in the brain. To dissociate the fear from the environment, the subject is exposed to the environment in the absence of any fearful stimuli until the environment is no longer predictive of any fearful outcome. This is referred to as extinction learning, and is thought to form a second memory in the brain which inhibits the original fear memory. Surprisingly, after successful extinction learning, animals are still prone to fear relapse under certain circumstances, which seems to undo the extinction learning. Whether this relapse is accompanied by a re-emergence of the original fear memory had not been well described. Using a combination of activity-dependent neuronal ensemble labeling, fluorescent microscopy with co-localization analysis, in vivo calcium imaging, and in vivo optogenetic perturbation, we discovered that the neuronal ensemble that is initially active during fear memory encoding re-emerges after fear relapse, and inhibition of the original fear ensemble can mitigate the fear response after relapse. This work was published in Neuropsychopharmacology.
Open Source Projects
I am a big proponent of the open-source scientific community, and I try to contribute to it whenever possible. Below are some code repositories meant to support the data collection, analysis, and visualization of several different kinds of data.
Publications & Preprints
BumJin Ko*, Madeline E. Bacon*, Yosif Zaki*, Denise J. Cai.
“Chemotagging: A chemogenetic approach for identifying cell types with in vivo calcium imaging”
bioRxiv, 2024. PMID: 39651295. in revision. 2025.
“Chemotagging: A chemogenetic approach for identifying cell types with in vivo calcium imaging”
bioRxiv, 2024. PMID: 39651295. in revision. 2025.
Zachary T. Pennington, Alexa R. LaBanca, Shereen D. Abdel-Raheim, Madeline E. Bacon, Afra N Mahmoud, Patlapa Sompolpong, Austin M. Baggetta, Yosif Zaki, BumJin Ko, Zhe Dong, Alexander CW Smith, Paul J. Kenny, Denise J. Cai.
“An anterior hypothalamic circuit gates stress vulnerability”
bioRxiv, 2024. in revision. 2025.
“An anterior hypothalamic circuit gates stress vulnerability”
bioRxiv, 2024. in revision. 2025.
Yosif Zaki, Denise J. Cai.
“Memory engram stability and flexibility”
Neuropsychopharmacology, 2024. PMID: 39300271.
“Memory engram stability and flexibility”
Neuropsychopharmacology, 2024. PMID: 39300271.
Zhe Dong, Yu Feng, Keziah Diego, Austin M. Baggetta, Brian M. Sweis, Zachary T. Pennington, Sophia I. Lamsifer, Yosif Zaki, Federico Sangiuliano, Paul A. Philipsberg, Denisse Morales-Rodriguez, Daniel Kircher, Paul Slesinger, Tristan Shuman, Daniel Aharoni, Denise J. Cai.
“Simultaneous two-color imaging with a dual-channel miniscope in freely behaving mice”
Science Advances, 2025. eadr6470. PMID: 40601747.
“Simultaneous two-color imaging with a dual-channel miniscope in freely behaving mice”
Science Advances, 2025. eadr6470. PMID: 40601747.
Yosif Zaki, Zachary T. Pennington, Denisse Morales-Rodriguez, Madeline E. Bacon, BumJin Ko, Taylor R. Francisco, Alexa R. LaBanca, Patlapa Sompolpong, Zhe Dong, Sophia Lamsifer, Hung-Tu Chen, Simon Carrillo Segura, Zoé Christenson Wick, Alcino J. Silva, Kanaka Rajan, Matthijs van der Meer, André Fenton, Tristan Shuman, Denise J. Cai.
“Offline ensemble co-reactivation links memories across days”
Nature, 2025. PMID: 39506117.
“Offline ensemble co-reactivation links memories across days”
Nature, 2025. PMID: 39506117.
Geoffroy Delamare, Yosif Zaki, Denise J Cai, Claudia Clopath.
“Drift of neural ensembles driven by slow fluctuations of intrinsic excitability”
eLife, 2024. 12:RP88053.
“Drift of neural ensembles driven by slow fluctuations of intrinsic excitability”
eLife, 2024. 12:RP88053.
Roxana E. Mesias, Yosif Zaki, Christopher A. Guevara, Lauren Friedman, Ayan Hussein, Karen Therrien, Alexandra R. Magee, Nikolaos Tzavaras, Pamela Del Valle, Mark G. Baxter, George W. Huntley, Deanna L. Benson.
“Development and cadherin-mediated control of prefrontal corticostriatal projections in mice”
iScience, 2023. 26(10):108002. PMID: 37854688
“Development and cadherin-mediated control of prefrontal corticostriatal projections in mice”
iScience, 2023. 26(10):108002. PMID: 37854688
Lingxuan Chen, Taylor R. Francisco, Austin M. Baggetta, Yosif Zaki, Steve Ramirez, Roger L. Clem, Tristan Shuman, Denise Cai.
“Ensemble-specific deficit in neuronal intrinsic excitability in aged mice”
Neurobiology of Aging, 2022. 123, 92-97. PMID: 36652783
“Ensemble-specific deficit in neuronal intrinsic excitability in aged mice”
Neurobiology of Aging, 2022. 123, 92-97. PMID: 36652783
Yosif Zaki, Nancy Holt.
“To Beat Extreme Heat, NYC Needs to Look to the Streets”
Gotham Gazette, 2022.
“To Beat Extreme Heat, NYC Needs to Look to the Streets”
Gotham Gazette, 2022.
Yosif Zaki*, William Mau*, Christine Cincotta*, Amy Monasterio, Emma Odom, Emily Doucette, Stephanie L. Grella, Emily Merfeld, Monika Shpokayte, Steve Ramirez.
“Hippocampus and amygdala fear memory engrams re-emerge after contextual fear reinstatement”
Neuropsychopharmacology, 2022. 47(11), 1992-2001. PMID: 35941286
“Hippocampus and amygdala fear memory engrams re-emerge after contextual fear reinstatement”
Neuropsychopharmacology, 2022. 47(11), 1992-2001. PMID: 35941286
Stephanie L. Grella, Amanda H. Fortin, Evan Ruesch, John H. Bladon, Leanna F. Reynolds, Abby Gross, Monika Shpokayte, Christine Cincotta, Yosif Zaki, Steve Ramirez.
“Reactivating hippocampal-mediated memories during reconsolidation to disrupt fear”
Nature Communications, 2022. 13(1), 4733. PMID: 36096993
“Reactivating hippocampal-mediated memories during reconsolidation to disrupt fear”
Nature Communications, 2022. 13(1), 4733. PMID: 36096993
Zhe Dong, William Mau, Yu Feng, Zachary T. Pennington, Lingxuan Chen, Yosif Zaki, Kanaka Rajan, Tristan Shuman, Daniel Aharoni, Denise Cai.
“Minian, an open-source miniscope analysis pipeline”
eLife, 2022. 11, e70661. PMID: 35642786
“Minian, an open-source miniscope analysis pipeline”
eLife, 2022. 11, e70661. PMID: 35642786
Abby Basya Finkelstein, Heloise Leblanc, Rebecca H. Cole, Troy Gallerani, Anahita Vieira, Yosif Zaki, Steve Ramirez.
“Social reactivation of fear engrams enhances memory recall”
PNAS, 2022. 119(12), e2114230119. PMID: 35286206
“Social reactivation of fear engrams enhances memory recall”
PNAS, 2022. 119(12), e2114230119. PMID: 35286206
Lingxuan Chen, Kirstie A. Cummings, William Mau, Yosif Zaki, Zhe Dong, Sima Rabinowitz, Roger L. Clem, Tristan Shuman, Denise Cai.
“The role of intrinsic excitability in the evolution of memory: significance in memory allocation, stabilization, and updating”
Neurobiology of Learning and Memory, 2020. 173, 107266. PMID: 32512183
“The role of intrinsic excitability in the evolution of memory: significance in memory allocation, stabilization, and updating”
Neurobiology of Learning and Memory, 2020. 173, 107266. PMID: 32512183
Yosif Zaki, Denise J. Cai.
“Creating space for synaptic formation – a new role for microglia in synaptic plasticity”
Cell, 2020. 182(2), 265-267. PMID: 32707091
“Creating space for synaptic formation – a new role for microglia in synaptic plasticity”
Cell, 2020. 182(2), 265-267. PMID: 32707091
Brianna Chen*, Nathen Murawski*, Anahita B. Hamidi, Emily Merfeld, Emily Doucette, Stephanie L. Grella, Monika Shpokayte, Yosif Zaki, Christine Cincotta, Amanda Fortin, Abigail Finkelstein, Steve Ramirez.
“Artificially enhancing and suppressing hippocampus-mediated memories”
Current Biology, 2019. 29(11), 1885-1894. PMID: 31130452
“Artificially enhancing and suppressing hippocampus-mediated memories”
Current Biology, 2019. 29(11), 1885-1894. PMID: 31130452
Patrick Davis, Yosif Zaki, Jamie Maguire, Leon Reijmers.
“Cellular and oscillatory substrates of fear extinction learning”
Nature Neuroscience, 2017. 20(11), 1624-1633. PMID: 28967909
“Cellular and oscillatory substrates of fear extinction learning”
Nature Neuroscience, 2017. 20(11), 1624-1633. PMID: 28967909